
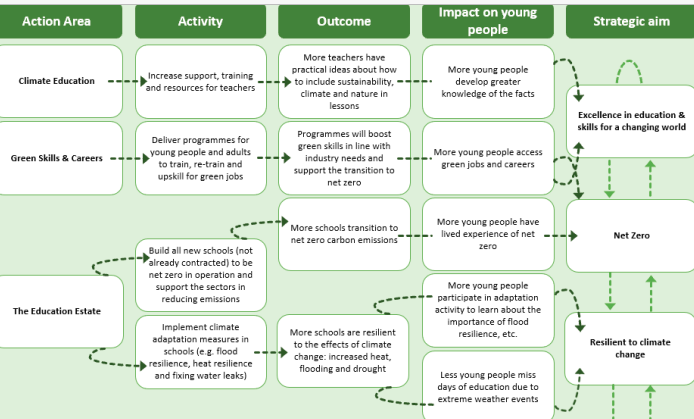
The United Kingdom is apparently about to incorporate the climate change agenda into nearly every aspect of public education. This is according to a paper released recently by the Government.
While the language sounds all lofty and altruistic, this comes across as a combination of propaganda mixed with child abuse. Terrorizing kids into believing the world is coming to an end is sick and cruel.
In fairness: this is still just a draft. However, it’s pretty disturbing that such things are being seriously discussed.
[Page 5] Context
This strategy sets out ambitious activity to respond to recommendations for education from the Committee for Climate Change, the Dasgupta Review, Green Jobs Taskforce report and supports the delivery of the Government’s 25 Year Environment Plan and Net Zero Strategy. It includes how we will work in the context of:
• The Paris Climate Agreement, which aims to hold the increase in global average temperature to well below 2 degrees Celsius above pre-industrial levels, and to pursue efforts to limit it to 1.5 degrees Celsius and includes measures relating to climate change education
• UK Government legislation to meet net zero by 2050. To achieve this, the UK is the first country to set legally binding carbon budgets, placing a restriction on the total amount of greenhouse gases the UK can emit over a five-year period. In the latest, Carbon Budget 6, the UK legislated to reduce emissions by 78% by 2035 compared to 1990 levels.
• UNESCO’s ‘ESD for 2030’ (Education for Sustainable Development) which sets out the key role of education in the successful achievement of the United Nation’s 17 Sustainable Development Goals.
• The Environment Bill which will deliver the most ambitious environmental programme of any country and drive forward action to protect nature and improve biodiversity, including through a target for species abundance for 2030, aiming to halt the decline of nature.
.
The UK requires the education sector to play its role in positively responding to climate change and inspiring action on an international stage.
If nothing else, the honesty is refreshing. The UK Government sees the “education” sector as vital in pushing the climate change narrative, achieving the Paris Agreement goals, and other environmental aspirations. The youth must be indoctrinated into accepting these goals.
[Page 12] Schools play an important role in preparing pupils for life in modern Britain and to become active citizens, whilst remaining politically impartial. Legal duties in this area help to ensure that pupils are allowed to form their own independent opinions on political issues that may define their future, without being influenced by the personal views of those teaching them.
.
Teaching about climate change and the scientific facts and evidence behind this, does not constitute teaching about a political issue and schools do not need to present misinformation or unsubstantiated claims to provide balance. However, in climate education there may be relevant political issues and partisan political views, for example on social and economic reform, that should be handled in line with schools legal duties on political impartiality. Importantly, whilst schools should support pupil’s interest in climate change and tackling both its causes and effects, it would not be appropriate to encourage pupils to join specific campaigning groups or engage in specific political activity, such as protests.
It’s interesting that presenting different viewpoints is considered “misinformation”. Now, limited discussion is permitted, but only in terms of what kind of solutions could be offered. In other words, the underlying claims of a crisis are not open for discussion. Is that how science works?
[Page 22] By 2025:
• DfE and its arms-length bodies will mandate that all suppliers bidding for contracts over £5m per year, commit to achieving Net Zero by 205012. Additionally, they must publish a Carbon Reduction Plan, which will clearly show how they will meet this target.
In the next few years, if a company wants to land a significant contract with the British Government, they’ll have to play along with this hoax, regardless of the costs to the business.
Documents like Agenda 2030 and the Paris Agreement are absolutely political in nature. To push them neutrally, without talking about the unending money flow is disingenuous. There really is no substitute for just reading the papers.
It’s infuriating, this technique of instilling fear and terror on impressionable minds. If you want society to stay intact, these are the measures that must be taken.
This review hardly does the paper real justice. The Government doesn’t even pretend that it’s not trying to trap the children at a young age.
Of course, it must be pointed out that Britain is hardly the only country to do this. Across the world, varying degrees of this indoctrination is going on. This is just some of the more obvious.
(1) https://www.gov.uk/government/news/education-secretary-puts-climate-change-at-the-heart-of-education–2#history
(2) https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1031454/SCC_DRAFT_Strategy.pdf
(3) UK Draft Education To Embed Climate Change Agenda Indoctrination
(4) https://canucklaw.ca/agenda-2030-un-sustainable-development-wealth-transfer-scheme/
(5) https://canucklaw.ca/ccs-2-the-paris-accord-a-giant-wealth-transfer-scheme/
(6) https://unesdoc.unesco.org/ark:/48223/pf0000374891
(7) https://www.gov.uk/government/publications/final-report-the-economics-of-biodiversity-the-dasgupta-review
(8) https://unesdoc.unesco.org/ark:/48223/pf0000374802
(9) https://www.oce.global/en
(10) https://www.oce.global/en/oce/partners
